
 Natural and Recycled Mineral Fillers in Rubber: Advantages and Limitations of Elastomeric Compounds
Natural and Recycled Mineral Fillers in Rubber: Advantages and Limitations of Elastomeric CompoundsHow CaCO₃, Talc and Recycled Mineral Fillers Influence Performance, Elasticity, Processability and Sustainability in Modern Rubber Compoundsby Marco ArezioIn the world of rubber compounding, the choice of mineral fillers plays a role that goes far beyond the mere balancing of compound cost. Every filler profoundly influences the rheology of the mix, its elasticity, its resistance to dynamic deformation, its softness, its surface appearance and, above all, its ability to withstand mechanical stress over long periods of use.This is where the most common natural mineral fillers used in the industry come into play: calcium carbonate (CaCO₃) and talc. Their presence in rubber formulations reflects a long industrial tradition, but one that carries well-known strengths along with equally well-known limitations.In recent years, however, the industry has begun experimenting with high-performance alternative fillers derived from advanced industrial processes. Among these, recycled mineral fillers stand out—based on iron, calcium, silica, magnesium and aluminum oxides—which, for simplicity, we will refer to as CR. These fillers originate from steel-processing by-products and feature exceptional purity, chemical stability, high hardness and an ultrafine particle size that makes them suitable even for elastomeric applications. The introduction of CR opens new technical possibilities, especially where the deficiencies of CaCO₃ and talc become limiting factors for elasticity and long-term durability.Natural fillers: why they are used and what advantages they offerCaCO₃ has always been one of the most widely used fillers in rubber thanks to its availability, low cost and ability to improve compound processability. Its presence facilitates extrusion, increases dimensional stability and provides a smooth and uniform surface appearance. It is particularly appreciated for general-purpose technical items, non-structural gaskets, elastomeric caps, soles and products where extreme mechanical resistance is not required.Talc, thanks to its lamellar structure, introduces a kind of “internal lubrication,” reducing friction during calendering or molding stages. Its inclusion improves flow behaviour, enhances surface appearance and promotes good shape stability, especially in EPDM, NR and SBR compounds.From an industrial perspective, the decisive advantage of these fillers lies in their low cost and their ability to extend the polymer matrix without excessively compromising processability. For standard applications, these properties are more than sufficient.The structural limits of natural mineral fillersHowever, CaCO₃ and talc have inherent limitations that cannot be overcome through compounding optimization alone. The first issue concerns elasticity: both fillers are non-reinforcing and introduce rigid points within the elastomeric matrix that interrupt the continuity of the polymer phase. In dynamic or highly stressed applications, this leads to reduced resilience, lower elongation at break and gradual weakening of the final product.CaCO₃ particles, especially if not perfectly micronized, can create stress-concentration points that act as triggers for micro-cracks. Talc, despite improving processability, further reduces the rubber’s ability to tolerate repeated deformation due to its plate-like structure, which promotes micro-fracture propagation along cleavage planes.Another often underestimated limitation concerns the natural variability of mineral fillers. Siliceous impurities, metallic residues and inconsistent particle-size distribution can negatively affect process stability, curing behaviour and the dynamic performance of the compound.Finally, from a strictly mechanical perspective, CaCO₃ and talc offer no true structural function: they do not increase tear resistance, do not improve heat resistance and do not enhance dynamic performance. For this reason, in more demanding applications they must always be combined with traditional reinforcing fillers.The technological shift: CR as an advanced solutionIn this context, the introduction of CR, a new-generation mineral filler derived from the controlled grinding of black slag from electric-arc furnaces (EAF), represents a turning point.This product offers unique characteristics:- high hardness (Mohs 7.5), far superior to CaCO₃ and talc- stable chemical composition (FeO, CaO, SiO₂, MgO, Al₂O₃ in constant ratios)- total absence of free silica, a crucial factor for operator safety- ultrafine particle size (
SEE MORE
 How to Improve the Molding of Non-Aesthetic Plastic Items
How to Improve the Molding of Non-Aesthetic Plastic ItemsConsiderations for the production and use of PO (PP/PE) granuleNon-aesthetic finished products, mainly intended for the market disposable, they were normally made with polypropylene compound, made up of a mixture of PP and PE (polypropylene + polyethylene) coming from the granulation of waste from separate collection. If we consider plastic pallets or spacers for rebar or fruit and vegetable crates, to give just a few examples, the mix between the two families of polymersmade it possible to produce compounds whose percentage of PP in the mixture could vary from 30-40% to 60-70%, depending on the envisaged recipe. The melting index at 230°/2.16 kg. it varied from 3 to 6 if the product had no added mineral fillers. The characteristics of the granule produced and, consequently of the final product, express a good performance with regards to resistance to compression and a less excellent in terms of flexural strength. As regards the ability to receive the colors in the extrusion phase of the granule or during the molding phases of the final product, yes may note that the medium-dark color range is the most appropriate, also by virtue of the fact that the base of the post-consumer semi-finished product to be transformed into granules is usually dark grey. Today the PO, which identifies the polyolefin mixture coming from separate waste collection, has taken on a different composition compared to the past due to the greater performance of municipal waste separate collection systems, which tend to maximize the removal of the polypropylene fraction from the PP/PE mix. This happens because the demand for polymers on the market tends to favor single compounds whether they are PP or HDPE or LDPE. The production trend described above involves having to work on a PP/PE mix which is qualitatively less performing than in the past, because they have been the balance between the three families, PP, HD, and LD which constituted the PO in the past has been altered. Furthermore, the increase in the production of both the waste to be processed and the demand for a granule from PP/PE compound has pushed some plastic waste treatment plants to accelerate the washing phase to recover productivity, decreasing the quality of the densified and ground material needed to produce the granule. We can list some critical issues in the production of PO compounds: • increase in LD% at the expense of HD in the polyolefin mix • worsening of the quality of the incoming wash due to the increase in volumes to be treated and the different % of polymers in the recipe • increase in the presence of bioplastics within the selected fraction which causes problems in the quality of the granule • increase in the use on the market of packaging made with mixed plastics involving a greater percentage of multilayer materials, such as some labels, which difficult to coexist with traditional PO . Compared to these changes in the basic composition of the PO and its processing , we will have to face problems to manage in the granule production phase and in the molding phase, in order to minimize the negative impacts of the quality of which the granule is composed. Regarding production, action should be taken: • on washing and drying times of the semi-finished product • on the size of the washing tanks • on water management (cleaning and replacement) • on the recipe of the PO compound for granulation • on filtration As regards the molding phase, the following should be done: • on machine temperatures • on the granule drying phase • on verifying mold cooling The technical intervention on these critical issues leads to the following improvements: • greater resistance to bending of the final product • improvement of aesthetic surfaces with reduction or disappearance of flashing on the finished product • improvement of color homogeneity • reduction of the bad odor of the granule and the finished product • increase in the life of screws and cylinders in the granulation phase and in injection molds • healthier workplaces during the plastic melting phases
SEE MORE
 Thermoforming of PET Sheets: Technology and Sustainability in the Production of Food Trays
Thermoforming of PET Sheets: Technology and Sustainability in the Production of Food TraysAn Analysis of the Thermoforming Process, Machine Structure, and the Use of Recycled PET for Sustainable and High-Quality Food Packagingby Marco ArezioThermoforming is a fundamental technique for producing food trays, and the use of PET (polyethylene terephthalate) represents an ecological and versatile choice. This article explores the functioning of thermoforming, describing the various stages of the process, the structure of the machines involved, and the important role of recycled PET.The Structure of the Thermoforming MachineThermoforming machines are complex and sophisticated, designed to transform plastic sheets into finished products through heat and pressure. A typical thermoforming machine consists of several essential components.The heart of the machine is the heating system. This can include infrared, electric, or contact heaters, which bring PET sheets to high temperatures, usually between 140°C and 160°C, making them malleable. It is crucial that the heating is uniform to ensure homogeneous and high-quality forming.Once heated, the sheets move to the forming station. Here, the PET is molded using a die. Forming can occur through vacuum, where a vacuum pulls the sheet onto the mold, or through pressure, which pushes the sheet into the desired shape. In some cases, a combination of both methods is used to achieve optimal results.After forming, the trays need to be separated from the residual sheet. This occurs at the cutting station, where blades or laser systems make precise cuts to obtain the final product. The excess material can be recycled and reintroduced into the production cycle, minimizing waste.Finally, the formed trays pass through a cooling system to solidify their shape. This cooling can be air or water-based, depending on the specific needs of the material and product.All these operations are managed by a central control unit, which monitors and regulates parameters such as temperature, pressure, and process speed. This ensures that each tray is produced with precision and consistency, maintaining high-quality standards.The Use of Recycled PETA significant aspect of using PET in thermoforming is the possibility of employing recycled material. PET is one of the most recyclable plastics, making it ideal for a circular economy.Recycled PET comes primarily from used bottles and other containers, which are collected, cleaned, and ground into small fragments. These fragments are then decontaminated and transformed into granules through an extrusion process. The obtained granules can be reformed into new PET sheets, ready to be used in thermoforming.The use of recycled PET offers numerous advantages. First, it reduces dependence on fossil resources and helps lower CO2 emissions. Additionally, using recycled materials helps reduce plastic waste, promoting environmental sustainability.Thanks to technological advancements, it is possible to produce recycled PET sheets with characteristics very similar to those of virgin material. This means that trays made from recycled PET can offer the same quality and safety as those made from virgin PET, making them suitable for food use.Moreover, it is possible to produce triple-layer PET trays, where non-food grade or non-virgin PET flakes are used in the central layer, while the outer layers can be composed of food-approved PET. This type of application helps reduce the production costs of PET trays.ConclusionsThermoforming PET sheets represents an advanced and versatile technique for producing food trays. The sophisticated structure of thermoforming machines and the well-defined process allow for high-quality products, essential for food packaging.The use of recycled PET, in particular, offers significant environmental benefits, contributing to sustainability and reducing the ecological impact of the plastics industry. This approach not only reduces waste but also promotes a circular economy, where materials are continually reused.In an era where sustainability has become a global priority, adopting techniques such as PET thermoforming and using recycled materials represent important steps toward a greener and more responsible future.
SEE MORE
 How to Verify Recycled Content in Plastic: The New Technology That Could Change European Packaging
How to Verify Recycled Content in Plastic: The New Technology That Could Change European PackagingHow Is the Percentage of Recycled Plastic in Products Really Measured: ISO Standards, European Rules, Supply Chain Audits, Mass Balance and Digital Watermarks in the New EU Packaging LandscapeAuthor: Marco Arezio. Expert in the circular economy, polymer recycling and industrial plastics processing. Founder of the rMIX platform, dedicated to enhancing the value of recycled materials and developing sustainable supply chains.Date: March 26, 2026Reading time: 16 minutesIntroductionSaying that a package “contains recycled plastic” is easy. Proving it in a serious, repeatable and defensible way before customers, authorities, auditors and the market is much more difficult. And today this difference matters more than before, because plastic is at the center of Europe’s new circularity policies: packaging accounts for about 40% of the plastic used in the Union and, in 2022, each European citizen generated 186.5 kg of packaging waste. The new European Packaging and Packaging Waste Regulation, the PPWR, entered into force on February 11, 2025, and its general date of application is set for August 12, 2026; among its objectives are increasing the safe use of recycled plastic and ensuring the recyclability of all packaging by 2030.The real question, therefore, is not only how much recycled plastic is in a product, but how it is actually proven. The correct answer is less intuitive than it may seem: in most cases, the percentage of recycled content cannot be read “by eye” nor certified with a single laboratory test on the finished item. Instead, it is built through a combination of regulatory definitions, mass balances, supply chain traceability, volume reconciliation, third-party audits and, increasingly, digital tools that improve the separation and qualification of incoming waste.What “recycled content” really meansThe technical basis starts with the definition. In the ISO field, recycled content is defined as the proportion, by mass, of recycled material present in a product. ISO 14021 remains one of the key references today for self-declared environmental claims and also includes the terms related to “pre-consumer material” and “post-consumer material,” that is, the distinction between material recovered before use by the final consumer and material coming from post-consumer waste.This point is decisive, because many market ambiguities arise here. A producer may declare 30% recycled content, but it is necessary to understand whether that 30% comes from internal or external industrial scrap, from urban post-consumer waste, from selected commercial waste, or from a combination of the two. From the point of view of environmental communication, the difference is not secondary: the technical quality of the material, the circular value of the claim and its market perception change significantly depending on the origin of the recycled content. ISO 14021 provides precisely this terminological and methodological framework to avoid vague or misleading claims.How the percentage of recycled plastic is calculatedThe basic principle is simple: it is a mass ratio. In the most straightforward case, the percentage of recycled content corresponds to the mass of recycled plastic incorporated into the product divided by the total mass of plastic considered within the scope of the claim, multiplied by 100. For single-use plastic bottles, the European Commission has already set specific rules: Implementing Decision 2023/2683 establishes that the proportion of recycled plastic is calculated by dividing the weight of recycled plastic in the bottles placed on the market by the total weight of the bottles placed on the market.But the formula alone is not enough. It is necessary to define the calculation perimeter precisely: batch, line, plant, annual period, product category, specific packaging family. It is also necessary to know which process losses were considered, which additives or masterbatches enter the formulation, and how the quantities coming in and going out are reconciled. Audit schemes based on EN 15343 and the most widely used traceability certifications require exactly this: documentary evidence, identification of flows and a plausibility check between inputs, yields, losses and declared output.Why the laboratory is almost never enoughHere we enter the heart of the problem. In theory, the laboratory is essential to identify the polymer, measure impurities, assess contaminants, verify MFI, ash, density, migration, odors or stability. In practice, however, the laboratory is almost never sufficient, on its own, to certify the exact percentage of recycled plastic contained in a finished product. The European Commission itself, in the section dedicated to the recycling of plastics intended for food contact, explains that the composition of recycled plastic cannot easily be subjected to official controls as happens with virgin plastic and that, precisely for this reason, controls focus on the production of recycled material and on audits of installations.The same approach also emerges from the technical literature of the European JRC in other highly regulated sectors: the verification of the content of recycled materials is described as based exclusively on documentation, with calculation rules, blending rules and measurement points defined upstream. In other words, the laboratory serves to qualify the material; the declared percentage, on the other hand, is demonstrated above all through chain of custody. This is an essential distinction for understanding why so many commercial declarations prove fragile when a robust traceability structure is missing.European traceability: EN 15343 as the cornerstoneIn the European context, EN 15343 is the cornerstone standard for recycled plastic. The standard specifies the procedures necessary for the traceability of recycled plastics and provides the basis for calculating the recycled content of a product. This means that the declared percentage does not arise from a qualitative perception of the material, but from a documented supply chain: origin of the waste, transformation, batch identification, internal controls, volume reconciliation and consistency between inputs and outputs.The certification schemes applied by the market move in exactly this direction. RecyClass, for example, explicitly states that its traceability certification verifies the exact percentage of recycled content through a controlled blending approach aligned with EN 15343 and ISO 22095; it also предусматриes third-party on-site audits and annual renewal of the certificate. This is important because it distinguishes a simple commercial self-declaration from an audited and defensible declaration.Mechanical recycling: the clearest case, but not a trivial oneIn mechanical recycling, the measurement of recycled content is generally more straightforward than in other scenarios. The recycled material enters as ground material, flakes or pellets; it is mixed with possible virgin material, additives or colorants; then it is transformed into the final product. In this case, the percentage can be demonstrated through a combination of purchase documents, supplier certificates, production sheets, compound recipes, mass balances and checks on the quantities actually processed, taking losses into account. Process audits require precisely a reconciliation of volumes to verify that the output corresponds to the recycled input used, considering yields, losses and additives.However, risks also exist here. If the incoming recycled material is not itself traceable or if it comes from poorly qualified heterogeneous streams, the numerical percentage may be correct on paper but weak in substantive terms. In other words, “50% recycled content” does not always have the same value: it matters whether it is truly traceable post-consumer PCR, pre-consumer industrial scrap, food-grade material, or a mixed stream with high qualitative uncertainty. For this reason, the strongest companies do not limit themselves to weighing the material, but document the origin and quality of the recycled content used.Food contact: when proof shifts even more toward the processIn food packaging, the issue becomes more rigorous. The European Commission points out that, when plastic is recycled for food-contact use, the problem is not only quantifying the recycled content but ensuring that any chemical contaminants have been removed to safe levels. Precisely because such contaminants may be unknown or variable, official control focuses not so much on the analysis of the finished product as on the decontamination process, good manufacturing practices and plant audits.This is also a crucial point for marketing communication. If a food container declares a certain recycled content, the credibility of that declaration depends not only on the numerical percentage, but on the ability to demonstrate that the recycled content was obtained within an authorized, monitored process suitable for the intended use. In food packaging, therefore, the “how much” and the “how” cannot be separated.Chemical recycling and mass balance: the most delicate issueWhen chemical recycling is involved, the matter becomes more complex because plastic waste is transformed into feedstock that is mixed with conventional raw materials in complex industrial systems. In these cases, the physical segregation of the “recycled” atom is not realistically practicable throughout the chain. For this reason, mass balance models are used, that is, chain of custody models that allocate a share of recycled content to outputs on the basis of accounting, temporal and allocation rules, without exceeding the quantity of recycled input that actually entered the system. ISCC PLUS describes this approach as one of the chain of custody options, alongside physical segregation and controlled blending.The issue is so central that ISO has also published ISO 22095-2:2026, specifically dedicated to the requirements and guidelines for applying the mass balance model in chain of custody systems. This is an important signal: mass balance is becoming less and less a mere “market” practice and increasingly an area of technical standardization.On the European regulatory level, the matter is still very much open. In July 2025, the Commission launched a consultation on new rules to calculate, verify and report chemically recycled content in single-use plastic beverage bottles. The proposed methodology is based on the fuel-use excluded allocation rule, meaning it excludes from recycled content any share of waste destined for fuels or energy recovery; it also provides for annual third-party verification for the most complex stages of the chemical supply chain and lighter requirements for SMEs. In February 2026, the Commission still indicated that it was in the final stage of defining these rules, not yet consolidated into a definitive and fully operational framework.The new technology that can truly change European packagingWhen people talk about recycled plastic, many imagine that there is a machine capable of taking a finished package, analyzing it and saying with precision: “there is 37% recycled plastic in here.” In industrial reality, that is not how it works today.The technology that can truly change European packaging is not a laboratory test capable of magically reading the recycled content of every package, but a system that helps separate packaging waste better before it is recycled. This system is based on digital watermarks, that is, small invisible or almost invisible codes printed on the package.To understand what this means, let us imagine a plastic tray for food, a detergent bottle and a cosmetic container. Today, when these packages arrive at a sorting plant, automatic systems can identify the type of plastic fairly well, for example PET, HDPE or PP, but they often have more difficulty distinguishing the original use of the package, that is, whether that plastic came from a food, cosmetic or household application. And this difference is very important, because plastics that appear similar may require different recycling pathways.This is where digital watermarks come into play. In practice, every package can carry with it a sort of “digital identity card” that can be read by sorting systems. This identity can tell the plant: “I am a food tray,” “I am a detergent bottle,” “I am a PP package,” “I belong to a certain category.” Thanks to this information, waste can be sorted much more precisely than with traditional systems.This is the real change: recycling is not improved at the end of the process, but at the beginning, when the waste is separated. If you start with a cleaner, more homogeneous and better classified stream, the recycled material obtained at the end will also be better.To make this even more concrete, one can think of the difference between collecting all fruit together in one large bin or separating it immediately by type and quality. If everything is mixed, the final result is a product that is less controllable. If, instead, separation is done properly at the origin, the final result is cleaner, more consistent and more suitable for quality uses. The same thing happens with plastic.This is why this technology is of such great interest to European packaging. Europe’s main problem, in fact, is not only recycling more, but recycling better. Much recycled plastic today has variable quality because it comes from waste streams that are too mixed and difficult to distinguish accurately. If sorting can be improved, the result is a purer, more stable and more reliable PCR, that is, post-consumer recycled plastic.This also has a very important consequence on the regulatory and commercial level. When a company declares that a package contains a certain share of recycled plastic, it must be able to prove it credibly. If the recycled material comes from a cleaner, traceable and well-separated supply chain, that declaration becomes more robust. In other words, digital watermarks are not used to directly “measure” the recycled content of the finished package, but to build a more reliable recycling chain, and therefore to make declared percentages more credible as well.From a practical point of view, their advantage is threefold. First: they help plants distinguish packaging better. Second: they make it possible to produce higher-quality recycled material. Third: they make it easier to connect that recycled material to serious supply chain documentation, useful for audits, certifications and compliance with the new European rules.So the central point is this: the technology does not change European packaging because it reads the recycled content already present in the product, but because it makes smarter, cleaner and more demonstrable recycling possible. And this is exactly what Europe needs today: not only more recycling, but recycling that can withstand technical checks, customer demands and future PPWR rules.What Europe is really asking for todayOn the regulatory front, Europe is moving on two levels. The first is the one already active for single-use plastic bottles: the SUP Directive requires 25% recycled plastic in PET bottles from 2025 and 30% in all plastic beverage bottles from 2030. The European Commission also recalls that in 2023 it adopted Implementing Decision 2023/2683 on the rules for calculating, verifying and reporting recycled content in single-use bottles.The second level is the broader PPWR framework. The official pages of the Commission clarify that the regulation entered into force on February 11, 2025, will generally apply from August 12, 2026, aims to make all packaging recyclable by 2030 and requires plastic packaging to incorporate increasing shares of recycled content with targets for 2030 and 2040. In other words, the verification of recycled content is no longer a niche issue for brands sensitive to sustainability: it is becoming a compliance infrastructure for the European market.How a company should really verify recycled contentIf a producer wants to avoid greenwashing and prepare for the new European context, it should not only ask itself “how much recycled plastic am I using?” but also “how will I be able to prove it in an audit?” The correct answer today is to build a system composed of four elements: a clear definition of the claim according to recognized standards; traceability of incoming material; mass balance with volume reconciliation; independent third-party verification when the market or customer requires it. This approach is consistent with ISO 14021, EN 15343, RecyClass schemes and the logic of European verification rules for bottles and food contact.In practical terms, a robust claim should specify at least three things: whether the recycled content is pre-consumer or post-consumer; which chain of custody model has been applied, that is, segregation, controlled blending or mass balance; which independent body has verified the system, if any. When this information is missing, the declared percentage may even be numerically correct, but it remains weak from an evidentiary point of view.ConclusionThe percentage of recycled plastic in products is not really measured with a single machine and is not demonstrated with an isolated formula. It is verified through an architecture of proof: ISO definitions, European traceability standards, mass balances, plant audits, supply chain documentation and, in the more advanced cases, digital systems that improve the separation and quality of recycled material upstream. This is the point that many commercial communications tend to oversimplify.The new technology that can change European packaging today is therefore not a “magic test” for reading recycled content in the finished item, but a technological ecosystem capable of making the supply chain more intelligent. Digital watermarks are probably the most concrete frontier in this direction, because they can increase sorting quality, create purer PCR streams and make future declarations about recycled content much more credible. In a European market that is moving from narrated sustainability to verified sustainability, this distinction will make the difference between those who communicate and those who prove.FAQHow is recycled content measured in plastic?As a rule, it is measured as the proportion by mass of recycled material in the product, but actual proof comes above all through traceability, mass balances and supply chain audits, not through a single test on the finished product.Is there a laboratory test that can say with certainty how much recycled plastic is in a package?In general terms, no: European sources show that verification of recycled content is based above all on documentation and process control, while final analysis alone is not sufficient to establish the exact declared share in all cases.What is the difference between pre-consumer and post-consumer?Pre-consumer comes from scrap recovered before use by the final consumer; post-consumer, on the other hand, comes from waste generated after use by households or commercial activities. ISO 14021 explicitly distinguishes these categories.What is mass balance in recycled plastic?It is a chain of custody model used above all when recycled and conventional feedstocks are mixed in complex systems, as in chemical recycling. In that case, the recycled share is allocated to outputs according to accounting rules that can be verified.Do digital watermarks measure recycled content?Not directly. However, they improve the separation of packaging waste and the creation of purer, more traceable streams, an essential condition for producing quality recycled material and making verification of recycled content in future products more robust.Real and verified sourcesEuropean Commission, Packaging waste and Packaging & Packaging Waste Regulation (PPWR), with data on entry into force, application date and objectives of the regulation.European Commission, Single-use plastics, with targets on recycled content in bottles and the chronology of implementing acts.European Commission, Plastic Recycling / Food Safety, with clarifications on controls, contaminants and the central role of process audits in food contact.ISO, ISO 14021 and ISO references on chain of custody and mass balance.European standard EN 15343, on the traceability of recycled plastics and the calculation of recycled content.European Commission, 2025 consultation on rules for chemically recycled content in bottles, with the fuel-use excluded method and third-party verification.AIM / HolyGrail 2.0 and HolyGrail 2030, on digital watermark technology and intelligent sorting results.ISCC PLUS and RecyClass, for chain of custody models, controlled blending, mass balance and traceability audits.Image under license© Reproduction Prohibited
SEE MORE
 What is the New Enzyme That Eats PET Waste in Quick Times
What is the New Enzyme That Eats PET Waste in Quick TimesWill the depolymerization of plastics through the new enzymes be the alternative to mechanical and chemical recycling? Today the production of plastic waste continues to exceed the capacity of their mechanical recycling, so much so that integrative solutions are being studied to reduce this gap. In addition to the countless avenues that could open up chemical recycling, biological engineering is making huge strides in identifying the correct enzymes that can degrade plastic. Through a study by a team of American scientists, aimed at identifying a modified enzyme, combinations of amino acids that could degrade PET in a shorter time were studied. faster than in the past. The organism has two enzymes that hydrolyze the polymer first into mono- (2-hydroxyethyl) terephthalate and then into ethylene glycol and terephthalic acid to be used as a source of power. One enzyme in particular, PETase, has become the target of protein engineering efforts to make it stable at higher temperatures and increase its catalytic activity. A team around Hal Alper of the University of Texas at Austin in the United States has created a PETase capable of degrading 51 different PET products, including containers and whole plastic bottles. In the construction of the study they used an algorithm that used 19,000 proteins of similar size and, for each PETase amino acid, the program studied their adaptation the environment they lived in compared to other proteins. An amino acid that doesn't fit well can be a source of instability and the algorithm suggests a different amino acid instead. Millions of combinations were then verified and, at the end of the analysis work, the researchers focused on three solutions that seemed to be the most promising ones. By further intervening with direct modifications, the scientists created a highly active enzyme on PET that worked quickly and at lower temperatures than in the past. At 50 °C, the enzyme is almost twice as active in hydrolyzing a small sample of a PET food container compared to another PETase engineered at 70 ° C. The enzyme even depolymerized an entire plastic cake tray in 48 hours, and the team showed it can create a new plastic item from degraded waste. It is important to emphasize that the tests were performed not on amorphous PET samples specially made in the laboratory, but on PET packaging purchased directly from supermarkets. This brings the tests performed even closer to the context in which they should operate, that is, in the context of recycling or depolymerization of plastics. It remains to be seen whether enzymatic depolymerization will eventually be used for large-scale recycling. In fact, most of the PET in the world is recycled not by depolymerization, but by melting and remodeling, but its properties deteriorate with each cycle. As we said there are some methods of chemical depolymerization, but they involve a very high energy consumption and, in view of the circularity of the products, the appearance of environmental impact that recycling entails must be taken into consideration, especially when renewable energies are not available. The great advantage of enzymes is that they can be much more specific than chemical catalysts and, therefore, it may be easier, in theory, to degrade a waste stream. Scientists do not hide, however, that the study of enzymes that depolymerize PET, however complicated and lengthy, could be even simpler than their applications on polyolefins or on mixed plastics. Automatic translation. We apologize for any inaccuracies. Original article in Italian.
SEE MORE
 Caprolactam and Polyamide: Chemical Composition, Environmental Impact and Strategies for Sustainable Production
Caprolactam and Polyamide: Chemical Composition, Environmental Impact and Strategies for Sustainable ProductionDiscover How Polyamides Are Produced from Caprolactam, the Chemical Components Involved, the Environmental Risks of Industrial Processing, and the Technologies for a More Sustainable Futureby Marco ArezioWhen we think of nylon—the well-known synthetic polyamide—we often associate it with textiles, strong ropes, precision gears, or lightweight, durable automotive components. Yet, we rarely pause to consider the intricate sequence of chemical reactions that makes its existence possible.Behind every strand of nylon lies an industrial chain built on aromatic compounds, controlled oxidations, acidic reactions, and a significant load of byproducts that raise serious concerns about sustainability.One of the key stages in this chain is the production of caprolactam, the cyclic monomer from which polyamide 6 (PA6) is formed, through a ring-opening polymerization reaction. Understanding the components required for its synthesis and the environmental implications of the process is not merely academic—it’s a necessary step to assess how the polyamide industry can evolve toward a more sustainable model.From Aromatic Molecules to Cyclic Monomers: A Complex TransformationIt all starts with benzene, one of the simplest and most stable molecules in aromatic organic chemistry. Primarily derived from petroleum fractional distillation, benzene serves as a feedstock for numerous processes in the heavy chemical industry.In the case of polyamide production, benzene is hydrogenated to form cyclohexane. This transformation takes place under high pressure and temperature in the presence of metal catalysts such as nickel or platinum, marking one of the earliest shifts from aromatic to aliphatic chemistry required for monomer synthesis.Cyclohexane is then oxidized to produce cyclohexanone, a cyclic ketone that plays a central role in the chain. This oxidation can occur either directly from cyclohexane with oxygen and metal catalysts or from phenol via selective reduction. The result is often a mixture containing cyclohexanol, known as KA-oil (ketone-alcohol oil). Cyclohexanone is separated from this mixture and purified, ready for the next transformation.Here enters a particularly reactive molecule: hydroxylamine, which reacts with cyclohexanone to form cyclohexanone oxime. Though chemically unstable, this compound is essential to the Beckmann rearrangement, a cornerstone of organic chemistry. In a strongly acidic environment—usually with sulfuric acid—the oxime is converted into ε-caprolactam, the cyclic monomer destined to become nylon.At room temperature, caprolactam appears as a white, crystalline solid, soluble in water. Compared to other precursors in the chain, it is relatively safe to handle. However, its production presents several critical issues.One of the most significant side effects of the Beckmann reaction is the generation of ammonium sulfate, a saline byproduct produced in nearly equal mass to the caprolactam itself. Although it can be used as a fertilizer, its disposal or reuse poses both logistical and ecological challenges.The Environmental Footprint of Chemical PrecursorsThe synthesis of caprolactam is far from harmless. Producing precursors such as benzene and hydroxylamine involves handling hazardous substances, performing energy-intensive reactions, and often generating polluting byproducts.Benzene, for instance, is a confirmed human carcinogen and requires strict safety protocols for its use. Hydroxylamine is unstable, highly reactive, and potentially explosive under certain conditions. Cyclohexanone, while less hazardous, is volatile and can contribute to atmospheric pollution in the form of volatile organic compounds (VOCs).One of the most concerning aspects is the emission of nitrous oxide (N₂O), a secondary byproduct that can emerge during various stages of the industrial process—particularly in the production of ammonia and nitrates used for hydroxylamine synthesis.N₂O is a greenhouse gas approximately 273 times more potent than CO₂, and it contributes directly to ozone layer depletion. In outdated facilities, producing just one ton of caprolactam can result in up to 9 kg of N₂O emissions.Toward a Sustainable Production: Challenges and ProspectsIn recent years, the chemical industry has made strides in reducing the environmental impact of caprolactam and its precursors. Some plants have installed catalytic abatement systems for nitrous oxide, cutting emissions by up to 98%. Others are experimenting with renewable feedstocks for cyclohexanone production, using biomass or sugars to reduce dependence on fossil fuels.In the realm of green chemistry, efforts are growing to develop bio-based caprolactam, using fermentation processes and intermediate synthesis from bio-derived adipic acid. However, these methods currently face scalability and competitiveness issues compared to well-established petrochemical routes.Another area of focus is the valorization of byproducts, especially ammonium sulfate, which can be used in fertilizer production. Yet from a systemic sustainability perspective, downstream reuse is not enough—upstream reduction in byproduct formation is essential, requiring a rethink of the entire production architecture.ConclusionThe production of polyamide 6 is a clear example of how a seemingly simple and ubiquitous molecule can hide a deeply complex chemical and ecological reality. From aromatics like benzene to reactive compounds like hydroxylamine, each step of the production chain involves technical decisions with significant environmental implications.To make the future of polyamides—and of engineered plastics more broadly—truly sustainable, it is not enough to focus on recycling the final product. Action must begin at the molecular level: rethinking monomer chemistry, choosing safer and less polluting precursors, and embracing technologies that minimize waste and emissions.The shift toward greener chemistry begins here—with a clear understanding of the molecules that shape our modern world.© All Rights Reserved
SEE MORE
 The Sustainable Future of Recycled LLDPE Granules
The Sustainable Future of Recycled LLDPE GranulesFind out how Advanced LLDPE Recycling Connects the Plastics Industry to the Circular EconomyLinear low-density polyethylene (LLDPE) is a plastic material widely used in numerous products, from flexible packaging to agricultural products. Its flexibility, strength and durability make it an ideal candidate for multiple applications.However, the massive use of LLDPE raises environmental concerns related to waste management and sustainability.The recycling of LLDPE, particularly in the form of recycled granules, emerges as a promising solution to address these challenges, reducing the environmental impact associated with the production and disposal of plastic materials.LLDPE Production ProcessLinear low-density polyethylene (LLDPE) is produced through a polymerization process that uses specific catalysts to achieve a unique molecular structure, which gives the material its desired characteristics of strength, ductility and flexibility.LLDPE production can be divided into several key stages:Linear low-density polyethylene (LLDPE) is produced through a polymerization process, as we have seen, that involves the joining of ethylene monomers into long polymer chains. This process typically occurs in two main ways:Gas phase polymerization: Gaseous ethylene is injected into a reactor containing a catalyst, usually based on titanium or chromium. High pressure and temperature conditions facilitate the bonding of ethylene monomers, forming the LLDPE polymer.Polymerization in solution or in slurry: Ethylene is dissolved in a solvent or suspended in a mixture in the form of a slurry, with the addition of a catalyst. The polymerization reaction occurs under these conditions, leading to the formation of LLDPE.The choice of process depends on the specific production needs and the characteristics desired in the final product. The LLDPE produced is known for its resistance to mechanical and chemical stress, flexibility and the ability to form high-quality thin films.Quality controlBefore being distributed, LLDPE undergoes rigorous quality control testing to ensure that the mechanical and chemical properties meet the standards required for specific applications.LLDPE Recycling ProcessLLDPE recycling begins with the collection and sorting of plastic waste, followed by a series of steps that transform the used material into recycled granules ready for a new production cycle:Collection and Sorting: LLDPE waste is collected and separated from other types of plastic and contaminants. This step is crucial to ensure the purity of the recycled material.Washing and Cleaning: The selected materials are washed to remove impurities, food residues, labels and adhesives. This process usually includes a shredding step, which reduces the material into smaller pieces, making it easier to wash.Regeneration and Pelletization: The clean LLDPE fragments are then melted in an extruder, which homogenizes the material. During extrusion, the molten material is filtered to remove any remaining contaminants. Finally, the melted LLDPE is cut into small granules, ready to be used in the production of new objects.This process not only reduces the amount of waste sent to landfill but also helps conserve natural resources and reduce the environmental impact associated with the production of new plastics.Environmental Benefits of Recycling LLDPERecycling LLDPE brings numerous environmental benefits, crucial to improving global challenges related to plastic waste and sustainability. Here are the main advantages:Reduced Carbon Footprint: Producing recycled LLDPE granules requires less energy than producing virgin LLDPE. This translates into a significant reduction in greenhouse gas emissions, contributing to the fight against climate change.Conservation of Natural Resources: Using recycled material reduces dependence on fossil resources, such as oil and natural gas, needed to produce ethylene, the basic monomer of LLDPE.Promotion of the Circular Economy: The recycling of LLDPE fits into the idea of a circular economy, where materials are reused and recycled as much as possible, reducing waste and improving resource efficiency.Applications of Recycled LLDPE GranuleThanks to its properties, the recycled LLDPE granule is used in various sectors:Flexible Packaging: Shopping bags, food packaging films and protective packaging materials are some examples where recycled LLDPE granules are used, taking advantage of its flexibility and resistance.Agricultural Products: Mulch sheets, irrigation pipes and protective nets are agricultural products manufactured from recycled LLDPE, benefiting from its durability and resistance to atmospheric agents.Consumer and Industrial Goods: Containers, waste sorting bins and industrial components are other examples of applications for recycled LLDPE, which enhance its resistance and versatility.Problems and LimitationsDespite the numerous advantages, recycling LLDPE presents some difficulties:Material Quality and Consistency: Variability in the quality of plastic waste can affect the properties of the recycled granule, sometimes making it difficult to use in applications requiring tight technical specifications.Technical Limitations: Some applications, especially those requiring material with specific mechanical or barrier properties, may be difficult to achieve with recycled LLDPE due to its variations in performance.Overcoming Technological and Market Barriers: To increase the use of recycled LLDPE, technological innovations that improve the quality of the recycled material and market strategies that promote its adoption are needed.Technological Innovations in LLDPE RecyclingTechnological innovations play a crucial role in improving the efficiency and effectiveness of the LLDPE recycling process, addressing issues related to the quality and applicability of the recycled material. Below are some of the main innovations in this field:Advanced Separation Technologies: New separation and purification methods, such as NIR (Near Infrared) technology and flotation techniques, allow for better material selection, increasing the purity of the recycled LLDPE and improving its properties.Compatibilization Processes: The development of compatibilizing additives helps to improve the interactions between different types of plastics in mixed recycling, improving the quality of the final product and expanding its possible applications.Quality Refinement through Chemical Recycling: Chemical recycling, which converts polymers into monomers or other intermediate chemical compounds, offers the possibility of producing recycled LLDPE with quality comparable to that of virgin, overcoming the limitations of mechanical recycling techniques.Market Trends and Impact of Environmental PoliciesThe recycled LLDPE market is influenced by several global trends and environmental policies:Growing Demand for Sustainable Materials: Growing environmental awareness among consumers and businesses is driving demand for recycled materials, including recycled LLDPE, especially in sectors such as packaging, where sustainability is becoming an increasingly important criterion of choice.Impact of Environmental Policies: Legislation and regulations, such as the European Union's circular economy directives, which impose quotas of recycled content in products and incentivize the reduction of plastic waste, stimulate the industry to invest in recycling and use of recycled materials.Supply Chain Innovations: Vertical integration and partnerships across the recycling supply chain are improving the efficiency of the recycling process and the quality of the recycled material, making recycled LLDPE more competitive in the marketplace.
SEE MORE
 Fiberglass: the difficult way towards a circular product economy
Fiberglass: the difficult way towards a circular product economyFiberglass: history, production, use and recycling. The difficult way towards a circular product economyFiberglass is certainly a product that has had a very important success given the flexibility of use, the relative ease of production and the technical characteristics of the manufactured products that could replace or improve the performance of other materials used up to then. Fiberglass was born in the 1920s, a period in which materials were being studied that had performance characteristics similar to those of construction metals (construction, aeronautics, naval) but which could add an advantage in terms of weight saving. During the 1940s, polyester was reinforced by using asbestos fiber, a composite plastic material with which, for example, additional tanks for aircraft were built . During the 50s of the last century, the increase in the production of glass fiber led to a progressive replacement of the asbestos fiber, creating technically more advanced products and widening the field of application. BUT WHAT IS FIBERGLASS? It is a glass reinforced composite plastic, also called VTR or GRP , using fabrics or felts with randomly oriented fibers and subsequently impregnated with thermosetting resins , generally liquid, composed of polyester or vinyl ester or epoxy , which harden and connect the fibers themselves through the action of catalysts and accelerators. The main characteristics of fiberglass products are: – Lightness – High mechanical characteristics – Durability – Corrosion resistance – Weather resistance – Excellent electrical insulation – Fire behavior manageable with specific additives – Good thermal insulation – Poor maintenance HOW ARE FIBERGLASS MANUFACTURED PRODUCTS? Premising that fiberglass is not a traditional plastic compound that needs heat and an important mechanical force (extrusion, injection, blowing) to make the products, but it is based on the work that the polymerized resin that comes into contact with the fibers does of glass. The main production processes are as follows: “Hand Lay-Up” consists in the coating by brush or roller of resins, correctly added with catalysts and accelerators, which determine their polymerization even at room temperature, on glass fabrics. The solidification of the resins allows the incorporation of the glass fibers present in the mold creating the article in vertroresina. “Filamnet Winding” consists in applying, on a normally metallic rotating cylinder, a wire impregnated with catalyzed resin. By winding this wire continuously on the mold, which will then be removed once the resin has hardened, cylindrical tubes or tanks can be created. “Resin Transfer Molding” consists in dry spreading, on one side of a mold, a set quantity of glass fibers, then the mold is closed again with its copy and the resin is injected, at low pressure, inside. With this system it is possible to carry out the injection procedure inside the mold also under vacuum. “Pultrusion” consists of a production similar to the classic extrusion of plastic materials, suitable for composite materials for the creation of particular profiles. WHICH SECTORS ARE INTENDED AND WHICH FIBERGLASS MANUFACTURES CAN BE REALIZED? The excellent technical and aesthetic qualities of fiberglass products allow them to be used in many fields with very wide applications: Railway sector Energy production Building do-it-yourself Nautical sector Sports works sector Electrotechnical market The products made with fiberglass are really many and it is not possible to mention them all, but we will indicate the products that, on the market, make the largest volumes: Hulls and articles for the nautical sector. Industrial and civil profiles Windows and shutters Skylights Covering sheets Walls Coatings for the refrigeration sector Stairs and walkways Coatings for the railway sector Coatings for civil transport Wind farms THE RECYCLING OF FIBERGLASS Fiberglass, being a compound material, as we have seen, escapes from the logic of the classic recycling of plastic materials , thus creating various and complex problems for its recycling. The first problem that we can remember is the presence of the thermosetting resins of which the product is composed, in fact, as we know, the polymerization reaction is always irreversible, this means that if we treated the ground fiberglass products with heat, as is done in generally with other plastics, we would not be able to return the resins used to liquid form. The second problem concerns the glass fibers that are used to reinforce the recipe . According to epidemiological studies conducted on animals in the laboratory, prolonged inhalation of the dust from these fibers would cause carcinomas and mesotheliomas. Although there is no evidence of human tests on animals, the European Community has issued a specific directive, including glass fibers among the dangerous substances subject to the labeling obligation. In fact, the glass fibers used for the manufacture of products are considered category 3 carcinogens and must bear the R40 label which identifies the possibility of irreversible health effects. Therefore, in the context of end-of-life recycling systems, we can report the main disposal destinations: Rubbish dump Grinding of powder products of various sizes and their reuse in sectors such as construction. Recycling by pyrolysis with the separation between fibers and resins Recycling by acid digestion Among the disposal systems used today, in terms of volumes, landfilling is certainly still the most used, with all the negative effects of the case. As for the grinding of powder products , it is certainly the easiest way, from a practical point of view, but leaves all the doubts from the health point of view that we have reported above open. While as regards recycling through pyrolysis or acid digestion it is not, today, economically convenient. It is clear that the way to dispose of the waste of fiberglass products, at the end of life, could be that of reusing ground powders in mixtures suitable for the production of finished products , but the volumetric reduction operation of fiberglass products must be carried out using suitable equipment , in isolated chambers, therefore not simple grinding mills, which safeguard workers’ health. There is also on the market a method of recycling fiberglass waste produced with orthophthalic, isophthalic or vinyl ester resins defined as “recovery with thermal-chemical treatment”. Through this process it would be possible to recover about 85% of the mother resin, in the form of a liquid and about 99% of the fibers that make up the reinforcement. Tests made by the manufacturer would show that the recovered resin, which is iodine-laden, could be mixed with the virgin resin for the creation of new products without there being any performance decay. As for the fibers recovered with this system, a calcination treatment is recommended on them, to eliminate the carbon residues present before being reused.
SEE MORE
 Micro Lamination of Plastic Films
Micro Lamination of Plastic FilmsMore and more thin, strong, elastic and easy to decorate, micro plastic films open new creative horizons. Polymeric micro films are now widespread in many areas, such as furniture, construction, protective products, automotive, food, packaging and many other fields. The formulation and production technology achieved by these laminates is allowing the exaltation of design and marketing through the transformation of products, not only from an aesthetic point of view, but also of technical performance. Actually in our imagination we place them in a recent time frame, but these thin plastic laminates can be dated to 1939, when they were first used as elements reflective in road signs. The applications, as we have seen, are really numerous and updated year after year, such as the films on the lenses of the glasses or on the visors of the helmets, times with writings on the outside that do not prevent you from seeing correctly from the inside. We can also mention the PVB films made with ink jet printing to be sandwiched between two glasses, so that the images become eternal as they are protected from glasses. Also interesting are the films for the preservation of food that can be purchased at the supermarket, there are those detectors, capable of highlighting deterioration or detecting the presence of GMOs. In the construction sector, for a long time now, polymeric films have been used to apply to glass to improve safety, reduce solar radiation that causes transmission of heat inside the premises with undoubted energy savings, reduce the refraction of light in order to darken the environments and shielded films able to reduce the introduction of more than 90% of electromagnetic waves inside the premises. In the field of safety, there are films composed of dozens of layers of very thin polyester, which are used to reduce the consequences of explosions. In fact, the elasticity that these polyester layers give to the glass, thanks to their elongation which can reach 150%, help the glass to better withstand the shock wave of an explosion. In the field of lighting and electronics, these micro films are designed to optimize the brightness of mobile phones, screens, computers, and for the transport of light itself . There are films on the market capable of reflecting or transmitting different wavelengths of light in the visible and infrared spectrum, consisting of hundreds of polymeric layers whose thickness they are of the order of a luminous wavelength. In these fields the science of physics of adjacent surfaces has made it possible to advance the technique of micro replication, the continuous repetition millions of times of a 3D microstructure consisting of tiny prisms or infinitesimal spheres invisible to the naked eye, which allows the creation of regular surfaces with specific properties, such as capturing the sunlight from different angles and distributing it inwards, or reflecting it totally outwards. Automatic translation. We apologize for any inaccuracies. Original article in Italian.
SEE MORE
 Static Mixers: Optimizing the Dispersion of Colored Masterbatches in Plastic Production
Static Mixers: Optimizing the Dispersion of Colored Masterbatches in Plastic ProductionThe Use of Static Mixers to Improve Uniformity and Reduce Costs in the Coloring Processby Marco ArezioIn the plastics industry, the quality and uniformity of the color of finished products are crucial to meet consumer expectations and maintain high production standards. However, inadequate dispersion of color masterbatches can lead to visible defects such as spots, streaks, or color shadows, compromising the quality of the finished product. This article delves into the use of static mixers as a solution to improve the dispersion of color masterbatches, especially in contexts where plasticizing capacity is insufficient. The Problem of Color Masterbatch Dispersion Color masterbatches are high concentrations of pigments or dyes dispersed in a carrier resin, used to color or impart other properties to plastic materials. Homogeneous dispersion of the masterbatch is essential to ensure the color uniformity and mechanical properties of the finished product. However, several factors, such as the viscosity of the polymer, the physical properties of the pigments, and the processing conditions, can negatively affect the dispersion, leading to production defects. Static Mixers: A Solution for Color Dispersion Static mixers represent an effective technology for improving the dispersion of color masterbatches without the need for moving parts. These devices utilize the geometry of their internal elements to divide, recombine, and orient the material flow to achieve homogeneous mixing. Unlike dynamic mixers, static mixers do not require external energy for movement, reducing operational and maintenance costs. Advantages of Static Mixers Improvement in Product Quality: The use of static mixers ensures optimal color dispersion, eliminating visual defects such as spots and streaks. Cost Reduction: The ability to achieve uniform dispersion with fewer quantities of masterbatch reduces the direct costs of materials. Versatility: Available for different processes, such as injection molding and extrusion, and adaptable to various types of resins and dyes. Sustainability: By minimizing the use of dyes and the generation of waste, static mixers help reduce the environmental footprint of plastic production. Key Components of the Static Mixer The Body of the Static Mixer: Structure and Materials The body of the static mixer is the structural element that encloses and supports the mixing elements, providing the channel through which the molten plastic material and color masterbatches pass during the mixing process. The design and construction of the mixer body are crucial to ensure efficiency, durability, and optimal integration into the production process. Key Features Resistance to High Temperatures and Corrosion: During the mixing process, the mixer body must withstand high temperatures and, depending on the materials treated, possible corrosive agents. This resistance is vital to maintain the structural integrity and functionality of the mixer over time. Chemical Compatibility: The material of the body must be compatible with a wide range of polymers and dyes used in the plastics industry, avoiding chemical reactions that could alter the properties of the finished product or damage the mixer itself. Internal Geometry: The internal geometry of the mixer body is designed to optimize material flow and facilitate effective mixing through static elements. This includes consideration of the flow channel shape, surface finish, and any specific features required for particular applications. Materials Used for Making a Static Mixer Stainless Steel: It is the most commonly used material for the body of static mixers in the plastics industry, thanks to its excellent resistance to corrosion, high temperatures, and compatibility with a wide variety of materials. Special Alloys: For applications requiring specific features, such as greater resistance to corrosion or temperature, special alloys can be used. Although more expensive, these materials offer superior performance in particularly aggressive environments. Coated and Composite Materials: In some circumstances, the mixer body can be made using composite materials or may be coated with specific materials to enhance resistance to corrosion, reduce material adhesion, or for economic reasons. Factors in Choosing a Static Mixer The selection of the static mixer body requires careful consideration of several factors: Production Process: The type of production process (e.g., extrusion or injection molding) can influence the choice of material and geometry of the mixer body. Material to be Processed: The chemical and physical nature of the polymers and dyes used determines the chemical resistance and thermal requirements of the mixer body. Operational Conditions: The specific conditions of temperature, pressure, and flow in the production process affect the selection of material and design of the mixer body to ensure optimal performance and durability. In conclusion, the body of the static mixer plays a crucial role in the success of the entire mixing process, directly influencing efficiency, product quality, and the durability of the system. Careful selection of material and geometry, based on a thorough understanding of the process needs and the properties of the materials handled, is essential to achieve the best results in the coloring of plastics. Energy Efficiency of Static Mixers in the Coloring of Plastics Energy efficiency is a crucial factor in the plastics industry, not only to reduce operational costs but also to minimize the environmental impact of production. Static mixers, thanks to their specific design and mode of operation, emerge as highly energy-efficient solutions in the coloring phase of plastic materials. Principles of Energy Efficiency No Moving Parts: Unlike dynamic mixers that require electric motors for the movement of paddles or blades, static mixers operate without any moving parts. This eliminates the need for additional energy to operate the device, significantly reducing the overall energy consumption of the mixing process. Optimization of Material Flow: The internal geometry of static mixers is designed to create an optimal laminar flow that ensures effective mixing without the need for additional mechanical force. This approach not only improves the quality of the mixing but also minimizes flow resistance, further reducing the energy needed to transport the material through the mixer. Integration into Existing Processes: Static mixers can be easily integrated into existing production systems without the need for significant modifications. Their passive operation results in minimal disruption to workflows and the ability to operate in synergy with the energy efficiency of existing facilities. Tangible Benefits of Using a Static Mixer Reduction in Operational Costs: The lower energy consumption of static mixers directly translates into reduced operational costs. This advantage is particularly significant in large-scale productions where even small efficiencies can accumulate substantial savings over the long term. Environmental Sustainability: Energy efficiency contributes to reducing the carbon footprint of the plastics industry. By using less energy, static mixers help companies move towards more sustainable production practices, in line with increasing regulatory pressures and consumer expectations for greater environmental responsibility. Reduced Maintenance: The absence of moving parts significantly reduces the need for maintenance and related production interruptions, indirectly contributing to energy efficiency. Fewer maintenance requirements mean less downtime and more efficient use of resources. Reducing Production Costs through the Use of Static Mixers The use of static mixers in the plastics industry offers significant advantages in terms of reducing production costs, especially in the coloring phase of materials. This section examines how static mixers contribute to reducing both direct and indirect costs, positively affecting the profitability of production operations. Direct Reduction in Material Costs Efficiency in Using Colored Masterbatches: One of the main advantages of using static mixers is their ability to disperse pigments from the masterbatch more uniformly and efficiently within the plastic resin. This efficiency allows achieving the desired shade using smaller quantities of masterbatch compared to traditional methods, leading to significant savings on material costs. Minimization of Production Waste: Uniform dispersion of dyes reduces the likelihood of visual defects such as streaks, spots, or color inhomogeneity in finished products. This translates into a smaller amount of production waste and, consequently, savings on costs related to re-melting, recycling, or disposing of defective materials. Optimization of Resources: The ability of static mixers to work effectively with different types of polymers and dyes allows companies to standardize mixing equipment, reducing the need for specialized devices. This aspect contributes to greater production flexibility and further cost containment. Indirect Reduction in Operational Costs Energy Efficiency: As previously mentioned, the absence of moving parts in static mixers significantly reduces energy consumption, leading to a reduction in operational costs related to energy. Reduced Maintenance: The simplicity of construction and the absence of moving parts in static mixers minimize maintenance needs. This reduces maintenance costs and downtime, improving the overall productivity of the plant. Durability and Reliability: The robustness and resistance to corrosion and high temperatures of the materials used for the bodies of static mixers ensure a long service life. The reduced need for replacement or repair further contributes to cost savings in the long term. Impact on Profitability The combination of these advantages—from reducing the consumption of raw materials to decreasing energy costs and maintenance—translates into a positive impact on the profitability of companies. Improved operational efficiency and superior quality of finished products can also strengthen the competitive position of companies in the market, attracting customers through the offer of high-quality products at competitive prices. Choosing the Right Static Mixer Choosing a suitable static mixer for the production of colored plastic materials involves a thorough assessment of various critical factors directly related to the production process itself. These factors influence not only the efficiency and effectiveness of the mixing but also the quality of the finished product, energy efficiency, and the reduction of production costs. Below, we examine the main critical factors for selecting a static mixer in relation to the production process. Type of Production Process The first critical factor concerns the specific type of production process in which the static mixer will be integrated, such as extrusion or injection molding. Each process has unique characteristics that influence the choice of mixer: Extrusion: Requires static mixers capable of handling continuous material flows and that can be effectively integrated into extrusion lines. The choice might lean towards mixers with a greater ability to manage material pressure and volume. Injection Molding: Here, the mixer must be able to handle intermittent production cycles with rapid changes in pressure and volume. A compact design that can be integrated close to the injection chamber might be preferable to minimize thermal degradation of the material. Materials to be Processed Selecting an appropriate static mixer for coloring plastics requires a deep consideration of the materials to be processed. This aspect is crucial because the physical and chemical characteristics of the polymers and color masterbatches directly affect the efficiency of mixing and the quality of the finished product. Below, we analyze the critical factors related to the material to be processed that must be evaluated when choosing a static mixer. Polymer Viscosity The viscosity of the melted polymer is one of the determining factors in choosing a static mixer. Materials with different viscosities require specific configurations of mixing elements to ensure homogeneous dispersion of the masterbatch: High Viscosity Materials: Require mixing elements that create wider flow channels or specific geometries to facilitate material movement and ensure effective mixing. Low Viscosity Materials: Can be processed effectively with narrower mixing elements that increase the interaction between the polymer and the masterbatch, improving color dispersion. Thermal Properties The thermal stability of the polymer and masterbatch is another critical factor. Heat-sensitive materials require a mixing process that minimizes exposure to high temperatures to prevent degradation. Selecting a mixer that ensures quick and efficient dispersion can help reduce the material's dwell time at high temperatures. Chemical Compatibility The chemical reaction between the material being processed and the static mixer, including its internal elements, can influence the choice of construction material for the mixer: Corrosion: Corrosive materials require a mixer built with corrosion-resistant alloys or advanced materials to avoid product contamination and mixer corrosion. Adhesion: Some materials tend to adhere to the internal surfaces of the mixer, requiring the use of materials or coatings that minimize adhesion to facilitate cleaning and maintain mixing efficiency. Particle Size and Shape of Masterbatch The size and shape of masterbatch pellets or particles can affect the mixing dynamics within the static mixer. Materials with different granulometries may require specific configurations of mixing elements to ensure uniform distribution of color in the melted polymer. Concentration and Type of Dye or Additive The concentration and type of dye or additive in the masterbatch determine the difficulty of achieving uniform dispersion and can influence the choice of mixer: High Concentration: Masterbatches with a high concentration of pigments or additives require more intense mixing to prevent clumping and ensure uniform color. Type of Additive: Specific additives may require particular mixing conditions, such as specific temperatures or mixing times, influencing the choice of mixer design and material. Production Capacity The desired production capacity can influence the size and design of the static mixer. Mixers with a larger internal volume or a specific arrangement of mixing elements may be necessary to handle high production volumes while maintaining the effectiveness of mixing. Integration into Existing Workflow The ease with which the static mixer can be integrated into existing production systems, without requiring significant modifications to the infrastructure or processes, is a critical factor. This includes considerations about the physical configuration of the plant, material flow logistics, and compatibility with other equipment. Environmental and Safety Considerations Finally, environmental and safety regulations can influence the choice of static mixers, especially in terms of the materials used, emissions, and energy consumption. Compliance with local and international regulations is essential to ensure sustainable and safe production. Operational Conditions of a Static Mixer Choosing an optimal static mixer for the coloring process of plastic materials must carefully consider the specific operational conditions in which the device will be used. These conditions can vary widely based on the type of production process, the nature of the materials handled, and the qualitative goals of the finished product. Below, we examine the critical factors related to the operational conditions that influence the choice of a static mixer. Process Temperature The temperature at which the polymer and masterbatch are processed is crucial for selecting the static mixer. Different materials require specific processing temperatures to ensure proper melting and mixing: Heat-Sensitive Materials: For polymers or dyes sensitive to high temperatures, it is necessary to choose a mixer that minimizes temperature increase during mixing, possibly through a design that promotes rapid heat transfer. High Melting Temperature Materials: Polymers that require high melting temperatures need mixers made from materials that can withstand such conditions without degrading or altering the properties of the product. Process Pressure The pressure under which the material is processed in the static mixer can vary significantly and has a direct impact on the selection of the device: High Pressure: Processes operating at high pressure require robust static mixers, capable of withstanding without deforming or losing mixing efficiency. Pressure Variations: Processes that experience wide fluctuations in pressure require a mixer designed to maintain consistent performance through these variations, ensuring homogeneous mixing regardless of pressure fluctuations. Flow Rate The speed at which material passes through the static mixer affects the quality of the mixing and overall production: High Flow: A high flow rate requires a mixer that can handle rapid volumes of material while maintaining uniform dispersion of dyes and additives. Low Flow Rate: For processes with slower flows, a mixer with mixing elements specifically designed to optimize contact between the polymer and masterbatch might be necessary to prevent material segregation. Available Space The size and configuration of the space where the static mixer will be installed play a significant role in selecting the device. It is essential to choose a mixer that fits into the existing infrastructure without requiring substantial modifications: Space Limitations: In environments with limited space, a compact mixer or one specifically designed to integrate into tight spaces may be necessary. Maintenance Accessibility: It's important to consider not only installation but also ease of access for maintenance or cleaning operations. Operational Duration The expected operational duration without interruptions is crucial for high-efficiency processes. Static mixers built with durable materials and designed for prolonged operations can reduce downtime and improve production continuity.
SEE MORE
 Polyacrylamide: Production, Applications in Polymers, Paper and Textiles and Recycling Perspectives
Polyacrylamide: Production, Applications in Polymers, Paper and Textiles and Recycling PerspectivesFrom Chemical Synthesis to Industrial Applications and Sustainability Challenges: A Journey into Polyacrylamide between Innovation and Circularityby Marco ArezioPolyacrylamide is one of the most versatile and widely studied synthetic polymers of recent decades.Produced from acrylamide, this substance has found widespread use in strategic sectors such as advanced polymer industry, paper manufacturing, and textiles. Its industrial history is closely linked to the search for increasingly high-performance materials, capable of meeting the demands of modern production without neglecting environmental issues related to sustainability and recycling.In this article, we will explore what polyacrylamide is, how it is produced, its most important applications in the polymer, paper, and textile sectors, and we will address the current challenges of its recycling from a circular economy perspective.What is Polyacrylamide?Polyacrylamide (PAM) is a water-soluble synthetic polymer obtained by the polymerization of acrylamide (CH₂=CHCONH₂), an organic compound derived from petrochemical processes. Its basic structure consists of a long chain of repetitive acrylamide units, with the possibility of modifying its chemical composition through copolymerization or variations in the lateral functional groups. This allows for a wide range of products, each with specific properties such as ionic charge, molecular weight, and water absorption capacity.One of the key features of polyacrylamide is its functional versatility: it can be produced in anionic, cationic, or neutral forms, depending on the intended application. This makes PAM extremely useful as an industrial additive, rheology modifier, flocculant, or binder.How is Polyacrylamide ProducedThe production of polyacrylamide begins with the synthesis of acrylamide, which can be obtained through catalytic hydration of acrylonitrile or through more sustainable enzymatic processes developed in recent years. The polymerization of acrylamide is then carried out in aqueous solution, using radical initiators (such as ammonium persulfate), and can be controlled to obtain different molecular weights and degrees of branching.The main production phases include:- Synthesis of acrylamide – via catalytic hydration of acrylonitrile, a process traditionally conducted on a large scale in the petrochemical industry.- Radical polymerization – acrylamide is subjected to polymerization in solution, suspension, or emulsion, in the presence of initiators, to produce long, linear, or cross-linked polymer chains.- Chemical modification (copolymerization or functionalization) – PAM can be modified during or after synthesis by adding ionic groups, functional molecules, or cross-linking agents to tailor its properties (for example, to increase affinity for specific ions or adjust viscosity).- Formulation of the final product – polyacrylamide is dried or packaged as a powder, beads, or concentrated solution.Continuous improvements in production processes aim to reduce the presence of residual acrylamide monomer, a toxic and potentially carcinogenic substance, thereby increasing the safety and sustainability profiles of finished products, especially for applications involving contact with food or people.Applications of Polyacrylamide in Polymers, Paper, and TextilesPolymer SectorIn the world of polymers, polyacrylamide is used both as a raw material for the production of advanced copolymers and as a functional additive. For example, copolymerization with other monomers (such as acrylic acid, acrylonitrile, diallyldimethylammonium chloride) allows for the creation of polymers with specific properties: greater hydrophilicity, surface charge, chemical reactivity, or thermal resistance.These copolymers are used in various applications, including:- Superabsorbent hydrogels (e.g., medical and hygiene sector): thanks to PAM’s ability to absorb large quantities of water and swell without dissolving.- Rheological additives and viscosity control agents: in paints, adhesives, drilling fluids, and personal care products.- Supports for chemical synthesis: as cross-linkers in resins or matrices for the separation of biomolecules in biochemical and biotechnology laboratories (polyacrylamide gel electrophoresis).Paper IndustryIn the paper industry, polyacrylamide is one of the most widely used additives to improve productivity and paper quality. It is employed as:- Retention agent: promotes the retention of fine fibers and fillers, reducing losses and increasing the yield of raw materials.- Drainage agent: accelerates water removal during sheet formation, optimizing production times and reducing energy consumption.- Mechanical properties enhancer: cationic polyacrylamide binds to cellulose fibers, increasing both the dry and wet strength of paper, as well as improving surface quality and printability.The use of PAM has enabled paper mills to become more efficient, lower operational costs, and reduce environmental impact by decreasing water consumption and the emission of residual sludge.Textile SectorIn the textile sector, polyacrylamide is mainly used as a thickener and binder in printing pastes and as an auxiliary in the treatment of wastewater generated by dyeing and finishing processes.The main functions include:- Thickener for printing pastes: improves the precision and definition of designs on fabric due to its ability to increase the viscosity of pastes without interfering with dyes.- Binder for fibers and pigments: promotes the adhesion of pigments or additives to fibers during printing or surface treatment stages.- Flocculant in purification treatments: allows for the effective removal of pollutants and suspended particles in wastewater, facilitating water recycling in textile processes.The result is improved final textile product quality, reduced consumption of raw materials, and greater sustainability of the production cycle.Recycling and Sustainability of PolyacrylamideThe issue of polyacrylamide recycling is complex and the subject of ongoing scientific research. As a highly stable and poorly biodegradable synthetic polymer, PAM does not easily lend itself to traditional mechanical or chemical recycling methods as do other more common polymers (such as PET). Nevertheless, studies are underway to find innovative solutions that minimize its environmental impact.The main strategies currently being examined are:- Recovery and reuse in industrial processes: in the paper and textile industries, sludges and residues containing PAM are partially recovered and reused as additives in other stages of the production process or as conditioners for sludge dewatering.- Advanced degradation: research is focusing on catalytic degradation technologies, advanced oxidation (UV, ozone, hydrogen peroxide), and biological methods using engineered microorganisms capable of attacking the polymer chain, although on an industrial scale these processes are still limited.- Development of biodegradable polyacrylamides: some companies are investing in the synthesis of copolymers with biodegradable segments or biopolymers that can replicate PAM’s functions but offer greater end-of-life sustainability.From a regulatory standpoint, the management of waste containing polyacrylamide is governed at European and national levels, with particular attention to minimizing residual monomer content and implementing safe recovery and disposal systems.ConclusionsPolyacrylamide is a key element in polymer chemistry and modern industrial applications, especially in the fields of functional polymers, paper, and textiles. Its ability to optimize production processes, enhance material properties, and facilitate water treatment makes it a valuable ally for sustainable production.However, challenges related to recycling and environmental sustainability require close attention to the development of new solutions to close the life cycle of these polymers and reduce the environmental impact of industrial processes.© All Rights Reserved
SEE MORE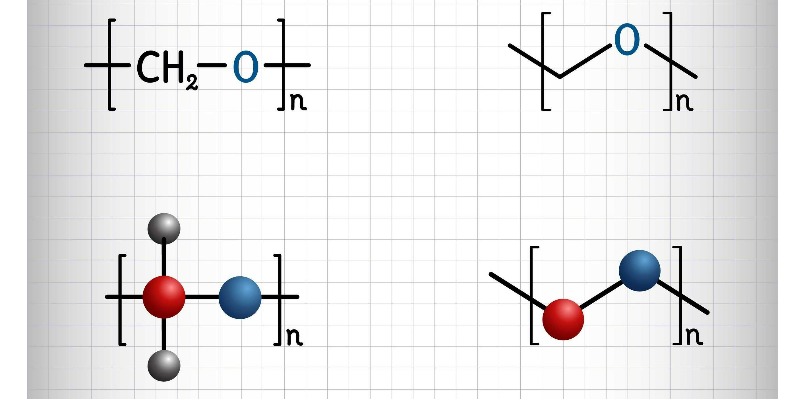
 POM or Recycled Polyoxymethylene: Where It Comes From and What It Is
POM or Recycled Polyoxymethylene: Where It Comes From and What It IsAcetal resin or paraformaldehyde (POM) is a recycled polymer with excellent technical characteristicsThe POM, commonly called alcetal resin, is a semi-crystalline polymer that is formed during the omo ( POM – H) or copolymerization (POM – R) of formaldehyde. The POM homopolymer, (CH2O)n, is among the stiffest raw materials, even in the absence of reinforcing elements, and has excellent dimensional stability. Polyoxymethylene or POM, is made up of a methylene bridge and an oxygen atom, which give the polymer high resistance and a low production cost compared to other polymers with similar mechanical characteristics. The main features of the POM are: • Good resistance to abrasion • Good resistance to high temperatures (up to 150° and constant up to 110°) • High surface hardness • Low coefficient of friction • Good electrical and dielectric insulation • Low permeability to organic substances, gases and vapors • Low resistant to strong acids (PH4) • Low resistance to oxidizing agents • Low resistance to UV rays if not additived • Low hygroscopicity • Not weldable at high frequency Processing of POM (Polyoxymethylene) The polymer can normally be treated with the usual processing systems of other thermoplastic materials, however the Injection molding is a widely used POM processing system. High molecular weight polymeric recipes normally lead to processing with extrusion systems, while slightly crosslinked ones are more suitable for blow molding. A precaution during the molding phases is to preheat the molds to a temperature between 60 and 130 °C, in this case the shrinkage reduces by 3 to 1% with decreasing mold temperature, and post-shrinkage increases proportionately. Fields of application of POM (Polyoxymethylene) By virtue of its performance characteristics in regarding toughness and hardness, products made with the POM polymer are suitable to the replacement of metal parts of technical use, such as levers, bearings, screws, gears, coils, pipe fittings, machine tool parts and pump components. Compound and blend with POM (Polyoxymethylene) The polymer lends itself to technical blends that can increase its strength and durability, in fact it is possible to add glass fiber, glass or mineral fillers. It is also possible to create blends between POM and PUR elastomers, these blends allow to increase toughness but, at the same time, decrease stiffness and strength, normally adding about 50% PUR elastomers. It is also possible to increase the dry frictional or sliding behavior by adding fillers of MoS2, PFT, PE or oils of silicone. On the other hand, aluminum or bronze powder can be added to the POM to increase heat stability and electrical conductivity. How to recycle POM (Polyoxymethylene) POM waste can be of an industrial or post-consumer type, however they are both valid products that can be recycled and used in technical blends. Industrial waste, which enjoys greater cleanliness at the outset, are generally pre-selected and subsequently ground, to then be used mixed with post-consumer ground or with virgin POM. This always depends on the type of transformation of the polymer that must be done and on the type of final product, both in terms of physical - mechanical characteristics and aesthetic appearance . Post-consumer waste needs greater attention during the recycling phase, in fact it may be necessary, after selection, a careful evaluation on the possible passage in a magnetic mill, to remove any metal parts, and a washing to separate the POM from non-metallic elements. Machine translation. We apologize for any inaccuracies. Original article in Italian
SEE MORE
 How a Recycled Plastic Bottle Is Formed
How a Recycled Plastic Bottle Is FormedExtrusion of the recycled polymer, formation of the parison, blowing of the bottle and detachment of the sprues The bottles of products for cleaning the house or for industrial liquids, made from recycled plastic, usually bought in shops, have had a huge spread in recent years, gradually replacing those made of glass and metal. They are undoubtedly lighter, have a lower production cost and are easily recyclable with a lower environmental impact than other liquid packaging. A bottle produced with recycled plastic can be produced, used, recycled and reused for a large number of times with a substantial saving of natural raw materials. But have we ever wondered how a plastic detergent bottle is produced? The recycling industry has made enormous strides by creating HDPE granules, the main polymer for bottles of liquid household cleaning products, which are increasingly performing and clean, which can be used 100% at least up to a volume of 5 liters of product. These polymers come mainly from the recycling of the bottles of the same detergents, through a careful selection of the collected waste and a series of operations to improve the secondary raw material, which allows the creation of another bottle with a thickness of a few microns. In order to produce a bottle in recycled HDPE, in addition to the polymer, we must have an extrusion and blow molding plant for the packaging. These systems are composed, in a very schematic way, of a feeder in which the HDPE polymer in granules will be placed, an extruder that will have the task of melting the plastic granule creating a moldable melt, a filter that will have the task, especially if a post-consumer recycled HDPE is used, to reduce as much as possible any pollutants present in the polymer and finally a mold in which the bottle is formed. Looking over the first part of the extrusion process, a topic already covered in a previous article, let's see what happens in the production process downstream of the extrusion. The HDPE melted by the extruder will be channeled into a plant suitable for the production of a tongue of plastic material, called parison, which will be the raw material for our future bottle. Once the quantity of material that constitutes the parison has been adjusted, the two parts of the mold will close together, imprisoning it. At this point, air will be blown into the parison, which will inflate the material on the walls of the mold, creating and cooling the bottle. The force with which the air is introduced is generally not higher than 10 Bar, allowing a correct formation of the product inside the mold, but the blowing duration depends on the volumetric size of the bottle to be made. Since this process is longer than the previous ones, it is possible to optimize the timing by using, for example, the exchange of blowing air to allow a faster phase cooling of the product inside the mold. As in all molding operations, even in the production of bottles it is possible that sprues are created around the raw bottle, which were once removed by hand. Currently the blow molding machines have special cutting edges which, automatically, trim the excess plastic present on the bottles, considerably speeding up the work. Once the bottle is formed, a conveyor belt will direct it to another automatic blowing system which will have the task, through the insufflation of air inside it, to check that there are no constructive imperfections, such as holes, which would compromise the seal once filled with product. After this control phase, the bottle can be suitable for the subsequent filling phase with detergents or other liquids to be marketed. Automatic translation. We apologize for any inaccuracies. Original article in Italian.
SEE MORE
 Moisture Absorption in Polymers
Moisture Absorption in PolymersTechnical Guide to Water Absorption in Plastics: Molecular Mechanisms, Industrial Drying, Hydrolytic Degradation, Recycling, Dew Point Control, and Management of Residual Moisture in PA, PC, PET, ABS, PMMA, PBT, PE, and PPAuthor: Marco Arezio. Expert in circular economy, polymer recycling, and industrial plastic processing. Founder of the rMIX platform, dedicated to enhancing the value of recycled materials and developing sustainable supply chains.Original article: May 2020Updated version: April 2, 2026Reading time: 12 minutesMoisture in polymers: why it is a decisive technical variableMoisture in polymers is one of those variables that often seems secondary until the production line begins to generate apparently inexplicable defects. Bubbles, surface haze, abnormal brittleness, opacity, viscosity fluctuations, dimensionally unstable parts, or sudden deterioration in finish are almost never random phenomena. Very often, behind these problems lies inadequate management of the water present in the material.Every polymeric material, during the stages of synthesis, packaging, transport, storage, and processing, comes into contact with environmental moisture. This interaction does not have the same meaning for all polymers. In some cases, water remains mainly on the surface; in others, it penetrates the pellet, diffuses into the material structure, and profoundly affects the behavior of the polymer during processing and in service.In the current industrial context, where companies must ensure constant quality, reduced scrap, lower energy consumption, and increasing use of recycled materials, moisture management can no longer be treated as a simple auxiliary step. It has become an integral part of process control, on the same level as temperature, residence time, plasticizing speed, and raw material quality.What hygrometric equilibrium means in plasticsAll plastics, to varying degrees, tend to reach equilibrium with the surrounding environment. This equilibrium depends on various factors: relative air humidity, ambient temperature, exposure time, pellet size, specific surface area of the material, the possible presence of dust or regrind, and the chemical structure of the polymer.Speaking of hygrometric equilibrium means describing the condition in which the material has absorbed or released water until it stabilizes with respect to the surrounding environment. This situation, however, is never absolute or final. A change in warehouse temperature, a seasonal shift, prolonged exposure to air, or a different storage method is enough to alter the moisture content of the material.It is also important to clarify a point that in the past was oversimplified: in hygroscopic materials, water does not always “chemically bind” in the strict and irreversible sense. More accurately, water molecules diffuse into the material and interact with specific polar sites along the polymer chains through intermolecular interactions, often in the form of hydrogen bonds. This distinction is relevant because it explains why a pellet may appear dry on the outside while still containing a significant amount of moisture inside.Hygroscopic and non-hygroscopic polymers: real differences in productionThe distinction between hygroscopic and non-hygroscopic polymers remains fundamental for those working in the plastics industry, but it must be understood technically and not merely in a textbook sense.In hygroscopic polymers, water penetrates into the pellet and distributes itself through the material by diffusion. This means that simply removing surface moisture is not sufficient. The polymer must undergo a drying treatment capable of extracting water also from the inner part of the pellet. Materials such as polyamide, polycarbonate, polymethyl methacrylate, polyethylene terephthalate, polybutylene terephthalate, and acrylonitrile-butadiene-styrene belong to this family in practical processing terms.In non-hygroscopic polymers, by contrast, water tends to remain mainly on the surface, without diffusing significantly into the matrix. This behavior is typical of polyolefins such as polyethylene and polypropylene, as well as polystyrene. In these cases, the problem is often linked to condensation, surface water, or moisture associated with poor storage conditions, rather than a true phenomenon of internal absorption.However, even this classification must be used intelligently. A material defined as non-hygroscopic is not automatically free from water-related problems. If the pellet has been exposed to moisture, if it comes from a washing cycle, if it has a high specific surface area due to grinding, or if it has been stored in cold environments and then exposed to warmer air, even a polyolefin can introduce into the machine an amount of water sufficient to generate defects.For this reason, in modern production language, it is no longer enough to ask whether the polymer is hygroscopic or not. It is necessary to understand where the water is located, in what quantity, how it got into the material, and what effects it can produce under the specific processing conditions.Why some polymers absorb water: polarity and hydrogen bondingTo truly understand the phenomenon of hygroscopicity, one must go down to the molecular level. The water molecule is polar: it has an asymmetric distribution of electric charges and is therefore capable of interacting with polar functional groups present in the macromolecular chains of polymers.The structure of the water molecule, with the oxygen atom being more electronegative and the two hydrogen atoms bonded at an angle of about 104.5°, generates a permanent dipole. Oxygen assumes a partial negative charge, while the hydrogens assume a partial positive charge. This polarity makes water particularly prone to interacting with other polar regions.When a polymer contains functional groups such as carbonyls, esters, amides, or other polar functions, the possibility increases that interactions with water molecules will occur. In polycarbonates, in polyesters such as PET and PBT, in PMMA, and in other technical families, the presence of the carbonyl group contributes to the polarity of the structure and facilitates the attraction of water molecules.In the case of polyamides, the phenomenon is even more evident. The presence of the amide group makes these macromolecules particularly sensitive to water. Water molecules can interact both with the carbonyl group and with the hydrogen bound to nitrogen, forming hydrogen bonds that promote the absorption and retention of moisture in the polymer matrix.Hydrogen bonds are interactions weaker than the covalent bonds of the polymer chain, but sufficiently stable to allow the adsorption and absorption of water up to a certain equilibrium value. This value changes from one polymer to another and is also influenced by environmental conditions. By contrast, non-polar polymers, such as many polyolefins, do not have a structure favorable to this type of interaction and therefore do not absorb moisture in the same way.What happens during extrusion and molding when the material is wetWhen plastic material enters the plasticizing barrel or the extruder, the presence of water becomes a critical factor. If the moisture is superficial, the first effect may be rapid evaporation during heating, with the consequent formation of bubbles, haze, opacity, silver streaks, craters, or microvoids. In these cases, the problem appears mainly aesthetic, but it can still compromise the commercial quality of the finished product.If, on the other hand, the polymer is hygroscopic and water is present within the pellet volume, the problem is deeper. During melting, moisture can promote hydrolysis reactions or, more generally, degradation processes that reduce the molecular weight of the material. This leads to a decrease in viscosity, a change in melt flow index, a loss of mechanical strength, and, in some cases, increased brittleness of the finished product.From a production standpoint, this means that the material no longer behaves as expected. The process technician may observe filling instability, pressure fluctuations, less stable cycle times, poorer dimensional stability, and defects that seem attributable to the machine or the mold, but which in reality derive from insufficient preparation of the raw material.The most common mistake is to consider moisture only as a defect of the material. In reality, it is a variable that alters the process. A wet polymer changes its rheological, thermal, and mechanical behavior, and therefore alters the entire balance of the transformation process.Hydrolytic degradation, surface defects, and loss of performanceOne of the most serious effects of moisture in engineering polymers is hydrolytic degradation. In the presence of water and high temperatures, some macromolecular chains may undergo scission. This phenomenon is particularly relevant in polyesters, polycarbonate, and other sensitive families, in which contact between residual moisture and processing temperatures can lead to a reduction in molecular mass.When molecular mass decreases, the material loses part of the properties for which it was originally selected. Toughness, impact resistance, the ability to withstand prolonged stress, and surface quality may all decrease. In many cases, the part may even appear visually acceptable but prove inferior under load, during laboratory testing, or in actual use.In polyamides, the relationship with water is even more complex. On the one hand, moisture in service can act as a plasticizer, increasing chain mobility and modifying stiffness, elongation, and dimensional stability. On the other hand, during processing, the uncontrolled presence of water can contribute to qualitative deterioration, which becomes especially pronounced when the material has already undergone other thermal histories, as happens in recycling or reprocessing cycles.This explains why the simple elimination of visible defects cannot be considered sufficient. A part free of bubbles is not necessarily a correctly processed part. True quality is measured by the preservation of the molecular structure and by the ability of the finished product to maintain the required performance over time.Industrial drying: hot air, dry air, and dew pointFrom a plant engineering perspective, moisture management is based on a distinction that remains valid today. In non-hygroscopic materials, where the problem is predominantly superficial, water removal can be carried out through hot-air dryers. In these cases, the goal is mainly to eliminate moisture adhering to the pellet surface or to prevent the effects of condensation.For hygroscopic materials, however, hot air alone is not sufficient. If the air introduced into the system still contains a significant amount of vapor, it will not be able to effectively extract water from inside the pellet. It therefore becomes necessary to use dehumidified air systems, in which the moisture content of the air is lowered before it comes into contact with the material.In this context, the concept of dew point becomes highly important. The lower the dew point of the process air, the greater its ability to absorb moisture from the polymer. For this reason, modern drying is not evaluated solely as a function of the set temperature, but rather by the relationship between temperature, residence time, air flow rate, dew point, and the integrity of the entire system.Another often overlooked aspect concerns reabsorption. A properly dried material can quickly take up moisture again if it is left exposed to the ambient air of the production department, if the hopper is not adequately protected, or if too much time passes between drying and processing. From this point of view, good drying depends not only on the quality of the dryer, but also on the discipline with which the material is handled and fed to the machine.In 2026, moreover, the subject of drying is closely linked to energy efficiency. Drying a polymer correctly is essential, but doing so inefficiently can significantly increase the industrial cost of the process. For this reason, the most advanced plants now seek a balance between drying quality, energy savings, and adaptation of parameters to the actual condition of the material.Moisture and recycled polymers: an even more important critical factorIf moisture management is already fundamental in virgin material, it is even more so in recycled material. Recycled polymers may show greater variability, a previous thermal history, higher sensitivity to degradation, and, in many cases, a surface more exposed to interaction with the environment.Recycled pellets or regrind may also come from washing, shredding, prolonged storage, or handling in environments that are not perfectly controlled. This means that their water content may be more variable and less predictable than that of first-supply virgin material.In polymers sensitive to hydrolysis, this condition is particularly critical. If the material has already undergone an initial thermal and mechanical cycle, its tolerance to further degradation may be lower. The presence of residual moisture, combined with processing temperatures, can therefore accelerate the loss of molecular weight and further worsen the performance profile of the recycled material.For those working in the circular economy, this is an essential point. Recycling does not depend only on the ability to recover material, but on the ability to preserve its technical value. If moisture is not rigorously controlled, a significant share of the recycled material’s potential can be lost already during the processing phase.How residual moisture is measured in plastic pelletsA modern production department cannot rely only on visual or tactile experience to assess whether a material is dry enough. Residual moisture must be measured or, at the very least, controlled through standardized procedures.At the technical level, there are reference standards for evaluating water absorption and moisture content in polymer materials. Absorption can be studied using specific standardized methods, while the quantification of residual moisture in pellets is often carried out with dedicated laboratory techniques. Among these, Karl Fischer titration remains one of the most reliable approaches when it is necessary to measure very low amounts of water with precision.In addition to laboratory controls, more and more plants use in-line monitoring instruments or indirect verification procedures based on dew point, residence time, feeding conditions, and process behavior. True quality, however, arises from the combination of measurement, experience, and organization.Knowing how much water is present in the pellet is important, but it is equally important to know when the material was dried, how long it remained exposed to the environment, and whether the transport system to the machine maintained adequate conditions. Without this overall view, the numerical value alone may not be enough.Correct strategies for managing PE, PP, PA, PET, PC, ABS, PMMA, and PBTFrom an operational point of view, polyolefins such as PE and PP mainly require attention to storage, condensation prevention, and the elimination of surface moisture. If stored correctly, these materials present fewer critical issues related to internal water absorption, but they can still generate problems when they come from washing cycles or humid environments.Polyamides, on the other hand, require much more rigorous management. Their strong affinity for water demands accurate drying, control of exposure time to air, and careful evaluation of the final dimensional and mechanical conditions of the part. PET and PBT, as engineering polyesters, must be processed with very low levels of residual moisture in order to avoid hydrolysis and loss of performance. Polycarbonate and PMMA also require careful preparation to preserve transparency, surface quality, and stability of the molecular structure. ABS, while not always reaching the same critical level as polyamide, must not be underestimated and still requires proper pre-drying.The best strategy does not consist in applying one general rule to all materials, but in building a protocol consistent with the polymer family, the type of plant, the pellet format, the possible presence of recycled material, the season, the humidity of the production area, and the quality objectives of the final product.Moisture in polymers is a much more complex issue than it may appear at first reading. It is not simply a matter of wet or dry material, but a phenomenon involving chemistry, diffusion, environmental equilibrium, drying technology, rheology, aesthetic quality, and preservation of mechanical properties.Hygroscopic polymers absorb water into their structure and require dehumidification systems and rigorous procedures. Non-hygroscopic polymers, although less sensitive to internal absorption, are by no means free from problems and must still be protected from surface moisture, condensation, and poor storage practices.In the contemporary industrial landscape, marked by growing attention to recycling, energy efficiency, and quality stability, moisture control has become a fundamental competence. Only by understanding the specific behavior of each polymer and by building a coherent process of drying, measurement, and handling is it possible to process plastic materials in a reliable, technical, and sustainable way.FAQWhat does it mean for a polymer to be hygroscopic?It means that the material is capable of absorbing water even داخل the pellet and not only on the surface. This requires a true dehumidification stage before processing.Do PE and PP need to be dried?In many cases it is sufficient to remove surface moisture, but if the material has been washed, poorly stored, or subjected to condensation, even these resins may require careful drying.Why is moisture dangerous for PET, PBT, and polycarbonate?Because during processing it can promote hydrolytic degradation, reduction of molecular weight, and deterioration of mechanical and optical properties.Do polyamides change behavior when they absorb water?Yes. Water can act as a plasticizer, modifying stiffness, elongation, dimensions, and the mechanical behavior of the material.Is recycled material more sensitive to moisture?Often yes, because it has a previous thermal history, greater variability, and may come from washing or less controlled storage stages.How is residual moisture controlled in polymers?Through standardized procedures, laboratory instruments, dew point control, verification of residence times, and monitoring of material handling along the entire line.SourcesScientific literature on water diffusion in polymer materialsTechnical standards for measuring water absorption in polymersTechnical standards for determining residual moisture in plastic materialsScientific studies on the hydrolytic degradation of engineering polymersTechnical publications on the processing of virgin and recycled polymersRegulatory documentation on methods for measuring moisture and water absorptionSEO Category: news – technical – plastics – recycling – polymers – moistureLicensed image© Reproduction Prohibited
SEE MORE
 Pfas emergency in Plastics and Packaging Is there a solution?
Pfas emergency in Plastics and Packaging Is there a solution?Pfas in plastics and packaging: they are chemical compounds not present in nature, not biodegradable and harmful to healthLike all the medals that are respected, also the Pfas, an acronym of the perfluoroalkyl and polifluoroalkyl substances, have their shiny side and their dark side. The chemical compounds of these families, which number about 4700, were created in the laboratory and widely used since the 1950s in the food packaging industry, in pesticides, in non-stick pans, in cardboard containers, in fire foams, in shampoos , in paints, in stain-resistant products and in many other applications. In plastics we find them in the form of elastomers (Vinylidene fluoride, Fluorurates in general, Tetrafluoroethylene) or in polymeric materials (Magnesium salt-sodium-fluoride of silicic acid). The advantages of these substances, applied to finished products, lies in their water repellency, oil-repellency and thermo-resistance , which allow us to make, for example, a waterproof jacket , not to stick an egg to the pan , not to get dirty mayonnaise or oily substances when we eat a sandwich filled with paper and not let our hands get dirty in the cinema when we eat popcorn. Their chemical bond composed of fluorine and carbon makes the resulting molecule an element irreplaceable today in industrial applications, but also makes it non-biodegradable and extremely dangerous, as it is odorless, tasteless and colorless. These characteristics allow it to be easily dispersed in water, soil and air, remaining to damage the environment and human health for a long time. The plants absorb the Pfas through the irrigation water, they give it to the fruits and the animals , of which they feed and thus, magically end up on our tables and in our body. From the health point of view, many studies have shown that the accumulation of these substances in the human body can favor spontaneous abortions, alter fertility, cause testicular, thyroid and kidney cancer. What are the means available today to defend ourselves from the sneaky pollution of the Pfas? At present there are not many: we can count on active carbon filters in which the porosity of the filtering carbon has shown a certain effectiveness in intercepting the Pfas, but it is not an effective system on all molecules. But once again, biochemistry could give us an answer to the problem as a team of American researchers has discovered a bacterium, called Acidimicrobium A6 , which would have the characteristic of breaking the bond between fluorine and carbon in Pfas. The bacterium was discovered in an American swamp and studied for a long time as a result of its ability to split ammonium, exploiting the iron present in the soil, without using oxygen. This named reaction, Feammox , was reproduced in the laboratory, after cultivating new strains of bacteria and subjecting the new families to other tests relating to the substances present in the waste water. After 100 days of cultivation in waters containing, among others, also the Pfas, it was noticed that the bacterium had the ability to break down the two main binders, fluorine and carbon, reducing them by 60%. The discovery could be interesting, not only in the liquid contaminated by Pfas, but also in the soils because the bacterium acts in hypoxic conditions, that is of poor oxygen.
SEE MORE
 Hydrophobic and Super-Hydrophobic Polymers: Innovations in Polymeric Coatings for Waterproof and Self-Cleaning Surfaces
Hydrophobic and Super-Hydrophobic Polymers: Innovations in Polymeric Coatings for Waterproof and Self-Cleaning SurfacesThe latest discoveries and technologies in hydrophobic polymers capable of making surfaces waterproof and self-cleaning, with applications in various industrial sectorsby Marco ArezioIn recent years, research on polymeric materials has made great strides, with particular attention to hydrophobic and superhydrophobic polymers.These materials, thanks to their unique properties of waterproofing and self-cleaning capabilities, are finding increasingly widespread applications in sectors such as the textile and aerospace industries.In this article, we will analyze the characteristics of these polymers, their working principle, the technologies used to develop them, and potential industrial applications.Hydrophobic and Superhydrophobic Polymers: Definition and Operating PrinciplesHydrophobic polymers are materials that repel water due to their particular chemical structure. This property is manifested when water molecules, instead of adhering to the material's surface, form spherical droplets that slide off.The effectiveness of this repulsion is measured by the contact angle between the water droplet and the surface: an angle greater than 90° is indicative of a hydrophobic material. This phenomenon is particularly important for applications where it is essential to keep surfaces dry and clean, reducing moisture formation and the adhesion of unwanted particles.Superhydrophobic polymers push this ability even further. These materials have contact angles greater than 150°, meaning that water is not only repelled but practically "bounces" off the surface.This phenomenon, often inspired by nature (for example, the lotus effect), results from the combination of surface microstructures and specific chemical characteristics. The lotus effect can be observed in nature on the leaves of the lotus plant, which, thanks to a combination of roughness and waxy composition, manage to keep the surface dry and free from impurities.This principle has been emulated in the design of superhydrophobic polymeric materials for various applications.Technologies and Innovations in Polymeric CoatingsThe production of hydrophobic and superhydrophobic coatings utilizes several advanced techniques, including:Thin Film Deposition: This technique allows the application of a thin layer of polymer on a surface. Fluorinated polymers, such as polytetrafluoroethylene (PTFE), are commonly used due to their excellent hydrophobic properties.Using physical vapor deposition (PVD) and chemical vapor deposition (CVD) methods, it is possible to obtain uniform thin layers that offer high water resistance. The use of these thin films allows control over the thickness and composition of the coating, achieving high-performance surfaces in terms of hydrophobicity and durability.Surface Nanostructuring: Creating roughness at the nanometric level is crucial to achieving high superhydrophobicity. Nanostructures trap air between the surface and the water droplet, reducing contact and enhancing the hydrophobic effect.Techniques such as laser etching and electrospinning are frequently used to create this surface structure. Laser etching allows for the creation of specific micro and nano-patterns that mimic natural surfaces, while electrospinning can be used to produce very fine fibers that increase surface roughness.These nanostructuring methods are often combined to maximize the superhydrophobic effect and ensure greater coating stability.Low Surface Energy Polymers: The chemistry of polymers plays a fundamental role in hydrophobicity. Polymers such as polydimethylsiloxane (PDMS) have low surface energy, which facilitates the formation of water droplets that easily slide off the treated surface.This type of polymer is often combined with nanostructuring techniques to achieve a more effective superhydrophobic effect. The low surface energy reduces the tendency of water to spread over the surface, which is particularly useful in applications where contact with liquids must be limited.Applications of Hydrophobic and Superhydrophobic CoatingsThe applications of hydrophobic and superhydrophobic polymeric coatings are becoming more widespread, thanks to their waterproofing and self-cleaning properties. These coatings not only improve the aesthetic appearance of surfaces but also offer significant functional benefits:Textile Industry: Fabrics treated with superhydrophobic coatings repel liquids and dirt, making them ideal for technical and sportswear.These fabrics not only keep the user dry but also reduce the need for frequent washing, with a positive impact on the environment. For example, waterproof jackets and trekking pants with superhydrophobic coatings can maintain their performance in extreme weather conditions, improving user comfort and reducing fabric wear.This technology also has significant implications in the medical sector, where hydrophobic textile materials can help prevent contamination and improve safety.Construction Sector: Coating construction materials such as concrete and glass with hydrophobic coatings helps protect surfaces from moisture and degradation.Building facades treated with these materials maintain a clean appearance over time, reducing maintenance costs. Additionally, these coatings can prevent the formation of mold and lichen, extending the durability of structures.Construction materials treated with superhydrophobic coatings can also improve frost resistance, preventing damage caused by water penetrating cracks and expanding during freezing. In this way, greater durability of buildings in environments subject to adverse weather conditions is guaranteed.Automotive and Aerospace: In the automotive industry, hydrophobic coatings are used to improve windshield visibility and reduce the accumulation of mud and dirt.In the aerospace sector, these coatings are essential for reducing the risk of ice formation on the exterior surfaces of aircraft, improving efficiency and safety. The use of superhydrophobic coatings on drones and unmanned aerial vehicles is being tested to reduce the weight caused by water accumulation and improve operational capabilities in adverse weather conditions.Engine components and wing surfaces can also benefit from these coatings, improving aerodynamics and reducing the need for frequent maintenance.Electronic Devices: Superhydrophobic coatings are also used to protect circuits and electronic components from water and moisture, ensuring greater device durability and reliability, especially in harsh environments.This is particularly relevant for wearable electronic devices and sensors exposed to the elements, which need moisture protection to function correctly. For example, sensors used in precision agriculture or IoT devices placed outdoors can greatly benefit from this technology, improving their robustness and reducing the need for frequent replacements.Challenges and Future ProspectsDespite significant technological advances, the production of durable superhydrophobic coatings still presents some challenges.The main challenge is long-term stability: many coatings lose their hydrophobic effectiveness due to mechanical wear or exposure to harsh environmental conditions. This loss of effectiveness limits the longevity and applicability of the coatings in many industrial contexts.Current research focuses on methods to improve the mechanical resistance and durability of these coatings.One of the most promising strategies is to use composite materials and combine polymers with inorganic nanoparticles. Nanoparticles can improve abrasion resistance and thermal stability, making the coating more durable even under intensive use conditions.Furthermore, nature-inspired research, drawing from phenomena such as butterfly wings or fish skin, is leading to the development of new surfaces that combine self-cleaning, anti-freezing, and antibacterial properties. These multifunctional surfaces could pave the way for a new generation of intelligent coatings capable of adapting to environmental conditions and providing tailored protection.Another promising research direction involves using eco-friendly materials for the production of hydrophobic coatings. Growing attention to the environment drives researchers to develop polymers and synthesis methods that are less harmful to the ecosystem while maintaining desired performance.This could lead to sustainable solutions that reduce environmental impact without compromising material performance.ConclusionsHydrophobic and superhydrophobic polymers represent an innovation of great interest in the field of advanced materials. The ability to make surfaces waterproof and self-cleaning opens up new possibilities in numerous industrial sectors, from textile production to the aerospace industry.Despite challenges related to coating durability and resistance, future prospects are promising, thanks to the continuous evolution of technologies and an increasingly bio-inspired approach. The combination of innovative solutions to improve durability and environmental sustainability will help make these coatings increasingly accessible and versatile, promoting their large-scale adoption.The adoption of hydrophobic and superhydrophobic materials in industrial settings could also revolutionize surface maintenance, reducing the need for periodic interventions and improving operational efficiency.With the continuous progress of nanostructuring techniques and the integration of new materials, we expect these coatings to become increasingly sophisticated, offering not only hydrophobic properties but also chemical resistance, UV protection, and self-repair capabilities. This wide range of functionalities will transform how we conceive surfaces, making them increasingly adaptable and high-performing.© Reproduction ProhibitedSourcesBarthlott, W., & Neinhuis, C. (1997). Purity of the sacred lotus, or escape from contamination in biological surfaces. Planta, 202(1), 1-8.Marmur, A. (2004). The lotus effect: Superhydrophobicity and metastability. Langmuir, 20(9), 3517-3519.Gao, L., & McCarthy, T. J. (2006). Contact angle hysteresis explained. Langmuir, 22(14), 6234-6237.Nosonovsky, M., & Bhushan, B. (2007). Hierarchical roughness and wetting of engineering surfaces. Microsystem Technologies, 13(3-4), 357-364.Zhang, X., Shi, F., Niu, J., Jiang, Y., & Wang, Z. (2008). Superhydrophobic surfaces: From structural control to functional application. Journal of Materials Chemistry, 18(6), 621-633.
SEE MORE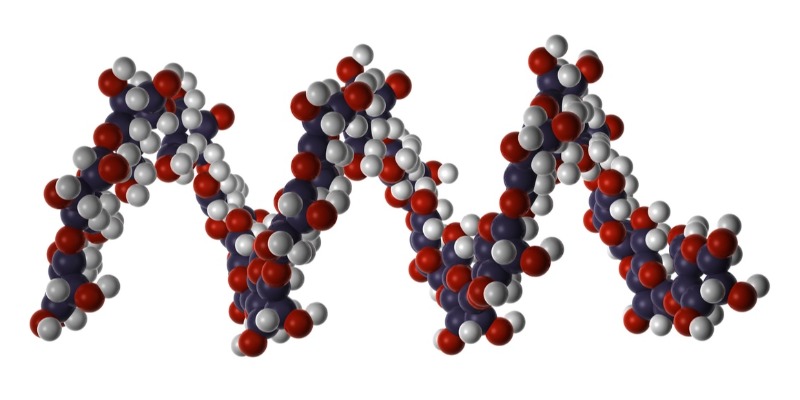
 The degradation of recycled polymers
The degradation of recycled polymersWhat is meant by degradation of recycled polymers: biological, oxidative, photo-degradation and thermal?In the years since the post-war period, plastics have increasingly taken the market by replacing products made with other types of materials as the countless advantages that this new material brought were immediately highlighted. Among the advantages of the plastic materials that can be underlined, we find the lightness, the ease of processing, the possibility of coloring and the low production cost. In fact, in those years we concentrated on the undisputed advantages of plastics without investigating the issues that determined their degradation . Today, with the great experience that users and manufacturers of plastics have acquired, we can balance the advantages and disadvantages of such an innovative material. We can classify the disadvantages between internal and external: Internal disadvantages chemical and physical modification polymer production process chemical reactivity of additives External Disadvantages thermo-hygrometric variations UV exposure pollutants heat microorganisms oxygen accidental causes Furthermore, degradation can be physical or chemical . In physical deterioration, an increase in crystallinity and consequently in density can be noted, with the emergence of internal tensions, cracks and deformations. The chemical one, which occurs at the molecular level, based on the degrading agent, affects the polymer chains with a loss of cohesion and a decrease in molecular weight.OXIDATIVE DETERIORATION Although the degradation of organic and inorganic polymers under the effect of oxygen is very slow, this causes the release of chemical substances that lead to the self-catalyzation of the polymer itself, that is, the chemical agents resulting from degradation in turn attack the polymer chain, activating a self-destructive process. Furthermore, if this phase is affected by the formation of free radicals by the action of heat or light , then the reaction between the polymer and oxygen increases the rate of cleavage of the chains, which leads to crosslinking and the formation of volatile elements. This process is called photo-oxidation or thermo-oxidation, depending on whether the trigger was light or heat. The direct consequences on the quality of the polymer can be seen through the reduction of the mechanical properties, especially with regard to elasticity and breaking strength. ORGANIC DECAY By biological degradation is meant the attack by fungi and bacteria on some polymers , especially those of natural derivation. These are subject to the phenomenon of hydrolysis , which can expose the polymer, in the presence of a high rate of humidity, to the breaking of the chains. To stop degradation, you can opt for storage in an oxygen-free environment, but you need to know the origin of the polymer well as it is not a universally valid treatment. THERMAL DETERIORATION The phenomenon of thermal degradation is caused by the presence of mobile hydrogens in the chain or by the radical activity that are triggered by heat, causing the chain to break with the formation of breaks and the production of volatile elements. The lack of oxygen leads to the depolymerization of the chain which occurs in three dissociative phases: initiation, molecular transfer and propagation. To increase the chemical resistance of the polymers to thermal degradation, the best solution is to add additives during production. PHOTO-DEGRADATION The photo-degradation phenomenon occurs when the polymer is subject to the influence of UV rays in the wavelength range between 290 and 400 nm. At the atomic level, we know that light radiation functions as a flow of particles, specifically photons , which, coming into contact with the molecules of materials and, under certain conditions, can interact passing from a state of low energy to one with high energetic excitation. . These particular flows and movements are defined as photo-physical and / or photo-chemical . In the first case, there are no chemical modifications between the polymer molecules, while for the photochemical process, there are possibilities that the molecules alter their chemical characteristic by virtue of the presence of abundant energy. In some synthetic macromolecules, the energy of the photons contained in UV radiation have the power to cause breakage of the covalent bonds. Automatic translation. We apologize for any inaccuracies. Original article in Italian.
SEE MORE